Helping sensitive cell therapies move between sites in a viable, ready-to-use state.
In Cell and Gene Therapy (CGT) workflows, transport is a process step with direct impact on viability and functionality. In autologous settings, this spans from patient-derived collection through ex vivo manufacturing to administration.
In allogeneic approaches, cells originate from established sources such as induced pluripotent stem cells or embryonic stem cells and undergo controlled expansion and differentiation prior to distribution.
Across both modalities, cells remain biologically active and sensitive to environmental conditions. As advanced therapies increasingly rely on fresh or non-cryopreserved material, warm chain handling becomes critical.
The Challenge
Across the CGT workflow, timing, temperature, sterility, and chain of identity directly affect product quality. Transport conditions must be tightly controlled to preserve viability, functionality, and lot-to-lot consistency across sites.
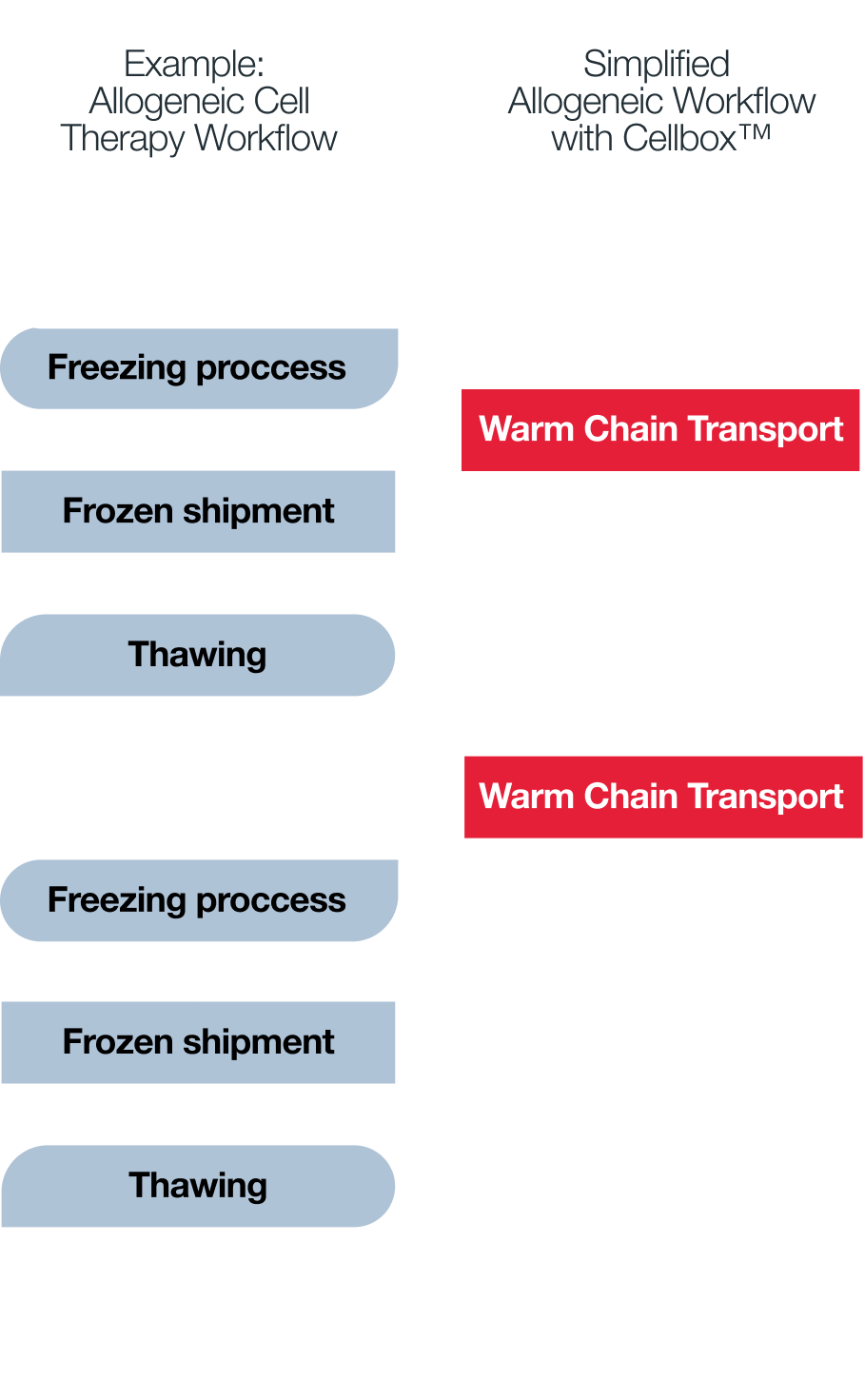
While some modalities such as CAR-T cell therapy can undergo cryopreservation, an increasing number of advanced cell types - including stem cell-derived therapies such as beta cells - show sensitivity to freezing. Cryopreservation can induce cell aggregation, reduce viability, and alter receptor expression, impacting functional potency.
Transport under physiological conditions, however, preserves native cell state, maintaining membrane integrity, receptor profiles, and metabolic activity - as shown, for example, for NK cells.
As more fragile and functionally complex therapies emerge, shifting from cryopreservation to controlled live cell transport becomes a critical factor for achieving consistent product quality and reliable clinical performance.
Why It Matters

Patient safety:
Viable, uncompromized cells enable safe and effective treatment
Therapy success:
Controlled transport preserves functionality and clinical performance
Cost & efficiency:
Fewer failures, delays, and product losses across the workflow
Where Cellbox™ supports
Across autologous and allogeneic CGT workflows, Cellbox™ supports controlled transport, preserving viability, functionality, and chain-of-identity from collection through manufacturing to administration.
From first-in-human to global rollout:
Across all phases, Cellbox™ supports controlled live cell transport – reducing complexity at every step.
Phase 1:
Local
5–20 patients
Single-site manufacturing and administration within one country. Simple logistics with no international coordination required. Allows focus on controlled transport conditions from the start.
Phase 2:
International Trials
~30–80 patients
Multi-site trials across borders no longer require building GMP infrastructure in the target country. Cellbox™ enables direct shipment from your existing facility – the product arrives ready for immediate clinical use.
Phase 3:
Full scale
~300–3.000+ patients
Global distribution demands standardized transport conditions, reproducible chain-of-identity, and full regulatory compliance across all participating sites and geographies.
NK Cell Study Live Cell Transport
“Live” cell shipment—a forward-looking transport option for cryo-sensitive cell-based therapies
» Live shipment delivered 2.5 times more functional NK cells after recovery than cryopreservation. «
Talk to our team
Talk to our team
If you would like to discuss your workflow or application, feel free to reach out.
Contact Form