Fraunhofer Institute for Cell Therapy and Immunology, Leipzig, Germany

Cell Therapy
“Live” cell shipment—a forward-looking transport option for cryo-sensitive cell-based therapies
January 2026
Lutz Roßbach¹, Thea Eichenberg¹, Nicole Pietzsch¹, Paul Franz¹, Stephan Fricke¹, Robin Sieg², Anna Dünkel¹, Kathrin Sabine Adlkofer²
² Cellbox Solutions GmbH
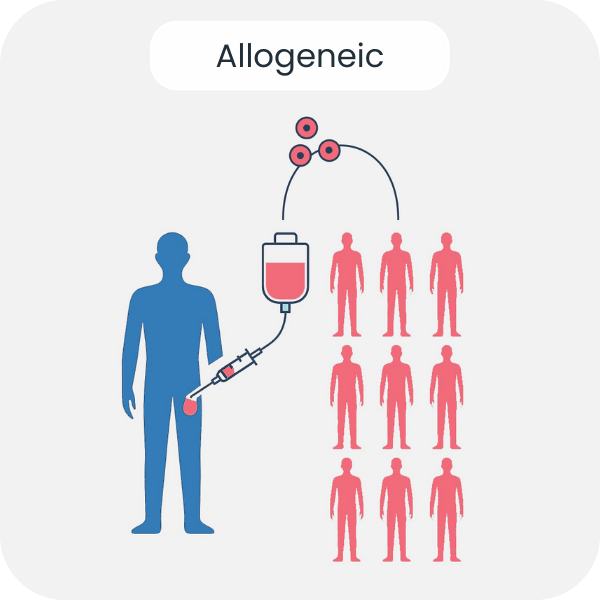
The study focuses on allogeneic NK cell products derived from healthy donors and highlights live shipment as a superior transport strategy for off-the-shelf NK cell therapies.
Cell therapies, which use the transplantation or manipulation of cells to treat diseases, are highly promising for conditions such as cancer, autoimmune diseases, and genetic disorders. Their success depends on the quality and viability of the administered cell products, which is especially critical for patients already weakened by chemotherapy or radiation. In such cases, ensuring high-quality cells that can engraft, proliferate, and function properly is essential for improving outcomes and survival.
Natural killer (NK) cells are strong candidates for cancer treatment due to their innate cytotoxic activity without the side effects of graft-versus-host disease (GvHD). However, cryopreservation has significantly hindered their clinical application, as it reduces both survival and function. Current “off-the-shelf” allogeneic NK therapies, therefore, face limitations because freezing and thawing lead to cell loss and impaired activity.
To address this challenge, we evaluated live shipment of NK cells using the Cellbox™ Shipper Flight, a transportable incubator maintaining 37°C and 5% CO2, versus cryopreservation and static laboratory incubation. Three independent NK cell batches, expanded under clinically compliant conditions, were tested. Optimized cryopreservation yielded 78% ± 9% of initial cell counts with >96% viability immediately after thawing. However, phenotypic analyses revealed a loss of NK markers, reduced cytotoxic activity, and an additional 50% ± 1% loss during 3 days of recovery, indicating that nearly half of the thawed cells were non-functional.

Live shipment was performed over approximately 44 hours using the Cellbox™ Shipper Flight.
In contrast, NK cells shipped live in the Cellbox™ retained an unaffected phenotype and cytotoxic function. After transport, cell counts recovered, and proliferation was observed during 3 days of static recultivation. Overall, live shipment delivered 2.5 times more functional NK cells after recovery than cryopreservation.
These findings demonstrate that shipping NK cells in culture using Cellbox™ is both feasible and advantageous, preserving cell function and ensuring a full therapeutic dose. By contrast, cryopreservation not only reduces immediate viability but also compromises long-term recovery and functionality, potentially limiting clinical efficacy. Mobile incubators like Cellbox™, therefore, represent a critical advance, enabling the reliable transport of living therapeutic cells and unlocking the full potential of cell therapies for patient benefit.
Preview: Figure 3
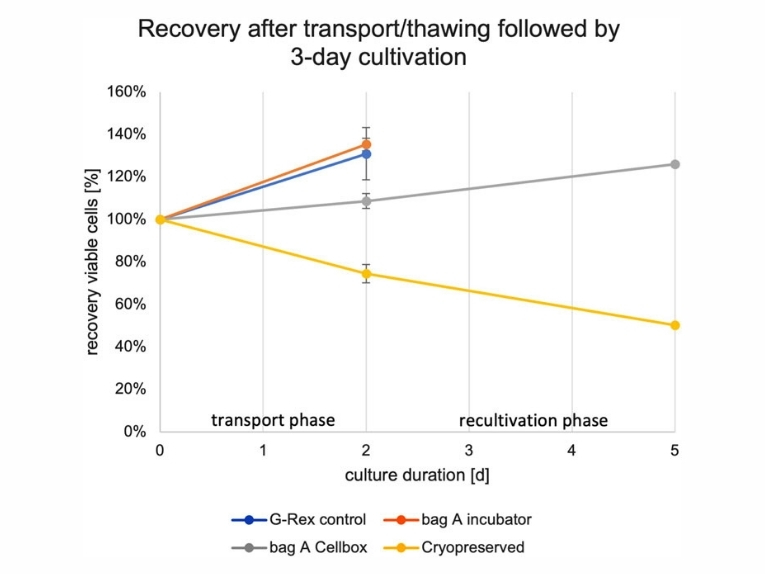
Figure 3. NK cell recovery after shipment (2 days) and recultivation phase (3 days) for G-Rex® [control] and Bag A stored in a static incubator, Bag A stored and transported in the Cellbox™, and cyropreserved NK cells. The 100% value is based on the seeded viable cell count at the start of the shipment phase for Donors 5 and 6.
Read the Full Paper here:
Frontiers in Bioengineering and Biotechnology
Edited by Guido Moll
Reviewed by Adam Joules & Raghavan Chinnadurai

